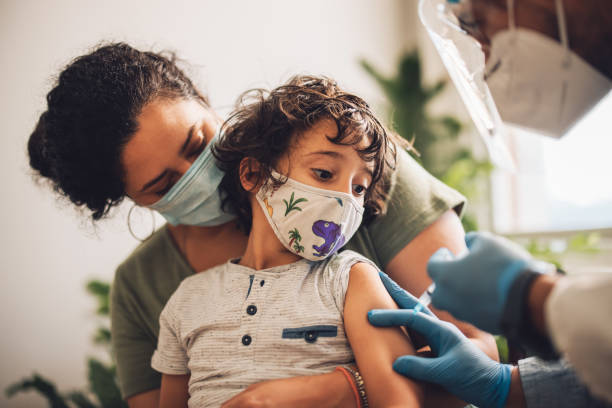
It is widely accepted that vaccination is key to bringing the COVID-19 pandemic under control by preventing severe disease. Due to the global disease distribution and diverse populations affected, multiple vaccine platforms are needed to overcome limitations in manufacture, logistics, and efficacy. Despite the global administration of up to 11·84 billion doses of COVID-19 vaccine to healthy adults and people at increased risk of disease, vaccinations for children are an emerging consideration.
In The Lancet Infectious Diseases, Krishna Mohan Vadrevu and colleagues provide initial safety and immunity data in children in India, who received a whole-virion adjuvanted inactivated SARS-CoV-2 vaccine (BBV152, Bharat Biotech International, Hyderabad, India). In Vadrevu and colleagues’ study, participants were enrolled into one of three groups (ages ≥2 to 6 years, >6 to 12 years, and >12 to ≤18 years), and monitored for solicited and unsolicited adverse events after vaccination. The vaccine is authorised for use in adults by WHO, although not by the US, UK, or EU regulatory authorities.
To grant licensure, regulatory authorities must consider the potential risks and benefits of vaccination. In paediatric populations, both sides of this equation are not yet fully understood. Risks that must be considered include patients at additional risk of severe disease, long COVID, and the potential for children to contribute to disease transmission to other vulnerable groups such as older people, or to teachers and caregivers (which could, in turn, lead to super-spreader events).
Controlled clinical trials are needed to inform benefits and risks of vaccination. Benefits might lie on a spectrum of reducing severe disease, reducing mild disease, or reducing disease transmission. The efficacy data on BBV152 in children are not yet reported, but neutralising antibody responses were comparable to those previously measured in adult populations.
The initial data indicate no serious adverse events, deaths, or withdrawals due to an adverse event, and no cases of myocarditis; however, ongoing monitoring in larger numbers of vaccine recipients are essential to identify rare occurrences of side-effects. Immediate reactogenicity (eg, pain at the injection site) was low, although limitations of the study include an absence of a placebo group and the small number of participants.
Most countries allow vaccination for children and adolescents aged 16–18 years, and several also approve vaccinations for children younger than 16 years.
Two other COVID-19 vaccines have been approved for children by the EU and WHO regulators, and both are mRNA based: BNT162b2 (Pfizer–BioNTech) and mRNA-1273 (Moderna). BNT162b2 is also approved by the US and UK regulatory authorities for use in children.
Where licenced, mRNA-1273 can be given to children aged 12 years and older at the same dose as adults. The European Medicines Agency has also authorised a half-dose to be given to children aged 6–12 years. BNT162b2 also requires a lower dose to be given to children younger than 12 years. Adults and adolescents older than 12 years receive 30 μg per dose, as opposed to 10 μg per dose for ages 5–11 years.
In this study, the BBV152 vaccine used two doses of 6 μg of whole inactivated virus with toll-like receptor 7/8 agonist molecules absorbed on alum, given in the same dose, volume, and regime as in the adult phase 3 clinical trial.
This strategy offers an easier mode of field implementation, because the same drug product can be formulated and stored to be used for both adults and children. This approach also reduces the potential for misdosing; however, vaccine safety and efficacy must not be compromised for the ease of manufacture or logistics.
Extensive assessment for paediatric clinical trials continues to be a challenge, mainly due to ethical considerations. Societal and behavioural attitudes towards vaccine acceptance are also important. The few demographic studies that exist show that during the COVID-19 pandemic, parents had increased hesitancy towards COVID-19 vaccination in children, probably associated with increased risk perception.
Knowledge around effectiveness and side-effects are crucial not just for national policy makers, but also for caregivers considering granting consent for their children to be vaccinated.
These trials are crucial to inform the cost–benefit analysis for policy and personal decisions. The risk and benefit of both disease and vaccination should be reported and discussed comprehensively among policy makers, because COVID-19 vaccination in children might assist recovery from the pandemic. But improving global accessibility to vaccines for vulnerable groups of adults, especially in countries with weak health-care systems, remains vital.
Article by:
article published in The Lancet
Tarikh Input: 14/07/2022 | Kemaskini: 14/07/2022 | m_radzi
PERKONGSIAN MEDIA